Table of Contents
Clinical trial portfolio optimization is the discipline of continuously evaluating, prioritizing, and resourcing a biotech firm’s active clinical programs against strategic objectives, probability of success, capital constraints, and competitive positioning. It’s what determines whether a company with five programs in the clinic and limited capital invests in the right three.
For biotech CTOs, this isn’t about project management in the traditional sense. It’s about building a decision architecture that connects the boardroom’s portfolio strategy to the clinical operations team’s daily execution. When the science changes, the portfolio view should change with it. When a Phase II readout disappoints, the resource reallocation should happen in days, not quarters.
The gap between knowing which program to prioritize and actually reallocating people, budgets, and timelines to reflect that priority is where most biotech firms lose capital and time.
The biotech execution gap: why strategy and operations disconnect
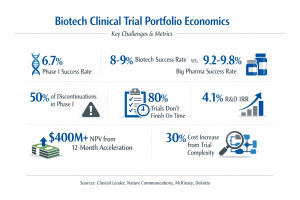
The data on biotech R&D productivity tells a sobering story. Phase I success rates have dropped to just 6.7% in 2024, down from 10% a decade ago (Clinical Leader / Evaluate, 2025). The biopharma industry’s internal rate of return on R&D investment has fallen to 4.1%, well below the cost of capital (Clinical Leader, 2025).
Biotech firms face this challenge with even fewer resources than large pharma. Nature Communications research published in 2025 found that biotech firms achieve overall clinical success rates of 8% to 9%, consistently lower than the 9.2% to 9.8% achieved by top-20 large pharmaceutical companies. The contributing factors include fewer development resources and capabilities and a higher risk appetite inherent in biotech business models (Nature Communications, 2025).
Why pipeline concentration amplifies the problem
With 23,000 drug candidates currently in development across the industry, hypercompetition in therapeutic areas is driving clinical trial costs higher (Clinical Leader, 2025). Oncology makes up nearly half of all R&D activity among the 20 largest biopharma companies, with the top five therapeutic areas accounting for 83% of programs (Deloitte, 2025). Clinical trial complexity is driving 30% cost increases (MedPath / Deloitte, 2025).
For a biotech firm with a concentrated pipeline of three to eight programs, each portfolio decision carries outsized risk. A single failed Phase II can redirect the entire company’s trajectory. Yet most biotech firms manage this concentration risk with disconnected spreadsheets, ad hoc executive reviews, and clinical operations systems that don’t talk to financial planning.
The cost of delayed decisions
McKinsey found that up to 80% of clinical trials fail to finish on time (McKinsey, 2024). For a company with three to five drugs entering first-in-human studies each year, a 12-month development acceleration applied across the portfolio can translate to more than $400 million in net present value (McKinsey, 2024).
Meanwhile, about 50% of assets discontinued by leading biopharma companies in both 2019 and 2024 were still in Phase I (McKinsey, 2025). The other 50% were discontinued in more expensive Phase II or III trials. Faster, earlier decisions about which programs to advance and which to stop would shift more of those discontinuations to Phase I, where the cost of failure is lowest.
Where the industry is heading
Portfolio prioritization is a continuous discipline, not an annual exercise
McKinsey’s analysis of leading biopharma portfolio strategies identifies three actions that top performers take: relying on therapeutic area expertise to inform investment decisions, expanding successful innovations across disease areas, and prioritizing external sourcing to enhance pipeline blockbuster potential (McKinsey, 2025).
The common thread is that portfolio prioritization must be robust and consistent, not something triggered by a cost transformation effort or acquisition. Biotech firms that treat portfolio review as a quarterly spreadsheet exercise miss the continuous signals from clinical data, competitive developments, and resource constraints that should reshape priorities in real time.
AI-driven trial design optimization
AI platforms can now identify drug characteristics, patient profiles, and sponsor factors to design trials more likely to succeed (Clinical Leader, 2025). Real-world data helps match patients more efficiently to clinical trials. These capabilities mean that portfolio decisions shouldn’t just be about which programs to fund, but how to design and execute trials that maximize the probability of success for each funded program.
Capital efficiency as a survival requirement
Between 2025 and 2029, an estimated $350 billion of revenue across the industry is at risk from patent expirations (Evaluate / Clinical Leader, 2025). For biotech firms competing for investment capital in this environment, demonstrating portfolio discipline and operational efficiency is no longer optional. Investors want to see that every dollar deployed connects to a quantified probability of clinical and commercial success.
How OnePlan connects portfolio strategy to clinical execution
OnePlan was named a Strong Performer in the Forrester Wave for Adaptive Project Management and Reporting, Q2 2024, with the highest scores for integration and product roadmap (Forrester, 2024). OnePlan is Microsoft’s PPM Partner of the Year for five consecutive years, with clients including Organon, BioMarin, and Johnson & Johnson.
Strategic alignment scoring for clinical programs
OnePlan’s strategic alignment scoring connects each clinical program to the firm’s investment thesis. When leadership defines priorities (therapeutic area focus, competitive window, probability of technical success), OnePlan scores every active program against those criteria and surfaces which programs are strategically aligned and which are consuming resources without matching the portfolio strategy.
Scenario modeling for portfolio trade-offs
When a Phase II readout shifts the probability of success for one program, OnePlan’s scenario modeling shows the downstream impact across the entire portfolio: how resource reallocation affects other programs’ timelines, what the financial impact looks like under different investment scenarios, and where capacity constraints create execution risk.
Resource demand planning across clinical operations
Biotech firms compete internally for the same clinical operations leads, biostatisticians, regulatory affairs specialists, and medical monitors across their portfolio. OnePlan’s resource demand planning shows capacity across all active programs, so staffing decisions on one trial account for commitments on every other active study.
Microsoft ecosystem integration
OnePlan integrates natively with Microsoft Teams for cross-functional clinical communication, SharePoint for regulatory document management, Power BI for portfolio analytics dashboards, and Dynamics 365 for financial planning. Clinical teams work within the tools they already use while leadership gets portfolio-level visibility without manual consolidation.
How Advaiya helps biotech firms implement portfolio management
Advaiya works with organizations across life sciences, pharma, and healthcare on project portfolio management implementations within the Microsoft ecosystem.
When Advaiya unified CRM systems for a Fortune 500 industrial manufacturer, the data integration challenges mirrored what biotech firms face with clinical portfolio management: 1M+ records across disparate systems, overlapping datasets, and the need for a single source of truth across 60+ countries. The results demonstrated what unified portfolio infrastructure delivers: 65% redundancy reduction, standardized processes across business units, and a single platform that eliminated the reconciliation overhead that kept teams in manual workarounds (Advaiya Case Study Compendium).
Advaiya brings enterprise architecture expertise that connects OnePlan’s portfolio capabilities to the specific way biotech clinical operations teams manage trial execution, regulatory milestones, resource allocation, and strategic investment decisions.
Connect with Advaiya about clinical trial portfolio management →
FAQs
Project management tracks individual trial execution. Portfolio optimization connects all trials to strategic priorities, resource capacity, and investment returns across the entire pipeline.
Yes. OnePlan tracks programs from preclinical through regulatory filing, with portfolio dashboards that show status, resource demand, and strategic alignment across all phases.
When a readout changes one program's outlook, scenario modeling shows the downstream impact on timelines, budgets, and resources across every other active program in real time.
OnePlan integrates with the Microsoft ecosystem and connects to external systems through APIs, allowing portfolio-level views that incorporate data from CTMS, regulatory, and financial systems.




