Table of Contents
Clinical trial portfolio management is the discipline of evaluating, prioritizing, and resource-planning across an entire drug development pipeline from discovery and IND filing through Phase I, II, and III trials to NDA/BLA submission and commercial launch. It’s not project management at the individual study level. It’s the strategic layer above: deciding which therapeutic areas deserve continued investment, which candidates to advance or terminate, and how to allocate constrained scientific talent and capital across competing programs.
For R&D leadership, this distinction matters because every portfolio decision cascades through years of execution. Advancing the wrong candidate into a Phase III trial doesn’t just consume $100M+ in direct costs; it ties up regulatory, CMC, nonclinical, and commercial teams for three to five years while stronger assets wait in the queue.
The economics are forcing ruthless prioritization
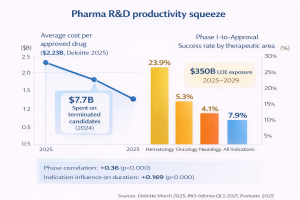
Deloitte’s 2025[1] analysis of the top 20 pharma companies found that the average cost to bring a single drug from discovery to market reached $2.23 billion in 2024, up from $2.12 billion the year before. Total development time now exceeds 100 months from Phase I to filing a 7.5% increase over five years (Deloitte, “Be brave, be bold,” March 2025).
Pipeline attrition is accelerating alongside those costs. The success rate for Phase I drugs reaching approval fell to 6.7% in 2024, down from 10% a decade earlier (Clinical Leader / Evaluate, 2025). Phase II the industry’s “valley of death” clears just 28.9% of candidates, the lowest success rate of any phase.
And the variance by therapeutic area is dramatic: hematology programs carry a 23.9% overall likelihood of approval from Phase I, while neurology sits at 4.1% and oncology at 5.3% (BIO, Informa Pharma Intelligence, QLS Advisors, 2021). A portfolio balanced across those areas can’t use uniform resource assumptions for any of them.
These failure economics aren’t theoretical. Deloitte’s[2] cohort spent $7.7 billion on clinical trials for candidates that were ultimately terminated in 2024 alone. Meanwhile, an estimated $350 billion of revenue faces loss-of-exclusivity risk between 2025 and 2029, forcing simultaneous management of patent cliff replacement and pipeline prioritization.
Where the industry is heading: AI-driven scenario planning and integrated demand forecasting
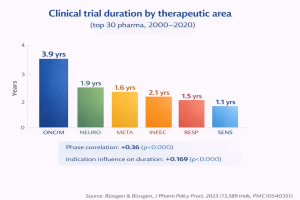
The traditional stage-gate model a go/no-go decision at the end of each phase, can’t absorb the complexity described above. An analysis of 13,589 clinical trials from the top 30 pharma companies found that study durations vary significantly by indication: oncology trials average 3.9 years versus 1.1 for sensory disorders, with higher phases correlating to longer durations at statistically significant levels (Büssgen & Büssgen, J Pharm Policy Pract, 2023). That variance means a portfolio spanning oncology, metabolic, and neurology programs needs distinct resource demand curves for each therapeutic area, not a single planning template applied across the board.
The Forrester Wave for Strategic Portfolio Management (Q2 2024) evaluated 12 providers across 22 criteria, reflecting the broader shift from simple project tracking toward integrated strategy execution. Forrester emphasized that removing information silos and enabling a holistic view for investment prioritization is now critical. AI-assisted portfolio tools that connect scenario planning with resource demand forecasting are becoming baseline expectations for organizations managing $100M+ R&D portfolios.
How OnePlan maps to pharma’s pipeline management reality
OnePlan is a strategic portfolio management platform built on Microsoft Cloud, recognized as a “Strong Performer” in the Forrester Wave Q2 2024, receiving the highest possible scores in integration and roadmap criteria. Microsoft has recognized OnePlan as a global PPM leader for five consecutive years. Pharma and biotech clients include Organon, BioMarin, and Generate: Biomedicines.
What makes OnePlan specific to life sciences rather than generic PPM is how it structures the pharma development lifecycle. Programs are organized by development stage, discovery through IND, Phase I/II/III, NDA/BLA submission, and commercial launch with functional sub-teams (CMC, nonclinical, clinical, regulatory, commercial) managed within each program. That structure means a portfolio view showing not just which assets sit in which phase, but what resource demand each asset creates across every functional team at every stage.
Three capabilities matter most for R&D leadership managing clinical portfolios. Scenario modeling lets teams compare portfolio configurations: what happens to your regulatory submission timeline if you advance two oncology candidates instead of one? What’s the resource impact of terminating a Phase II metabolic program and reallocating those CMC scientists? Resource demand planning surfaces capacity constraints before they become timeline delays, especially critical when specialized talent like biostatisticians and regulatory writers is shared across programs. And financial planning tracks estimated and actual spend by program, phase, and cost category, giving CFOs line-of-sight into R&D burn rate against quarterly forecasts.
OnePlan integrates natively with Microsoft Teams, Power BI, Azure DevOps, and Project for the web, alongside Jira, Smartsheet, and monday.com. Its Sofia GPT capability uses Microsoft OpenAI for AI-assisted resource forecasting and objective-setting, connecting execution-level tools to portfolio-level intelligence without requiring teams to change how they work day-to-day.
How Advaiya helps pharma teams implement portfolio management
Advaiya works with organizations across manufacturing, energy, and professional services on project and portfolio management implementations within the Microsoft ecosystem. When Advaiya deployed Dynamics 365 Business Central for a multi-entity consulting firm, the results reflected the same pattern effective portfolio management delivers: 80% improvement in billing accuracy and 60% reduction in approval dependency through centralized planning and integrated workflows.
For pharma organizations evaluating OnePlan, Advaiya brings enterprise architecture expertise, Microsoft integration depth, and implementation methodology that connects business process design to platform configuration, ensuring the tool maps to how your R&D teams actually make portfolio decisions.
Connect with Advaiya about pharma portfolio management.
FAQs
Trial management runs individual studies. Portfolio management prioritizes which trials to fund, staff, and advance across the pipeline.
Yes, it connects with Microsoft Project, Azure DevOps, Jira, Smartsheet, and monday.com without replacing study-level workflows.
Mid-size biotech implementations typically reach production use within 8–12 weeks, depending on data migration and team onboarding.
It manages milestones and deliverables by regulatory stage but isn't a regulatory information management system it connects those milestones to resource and financial planning.




